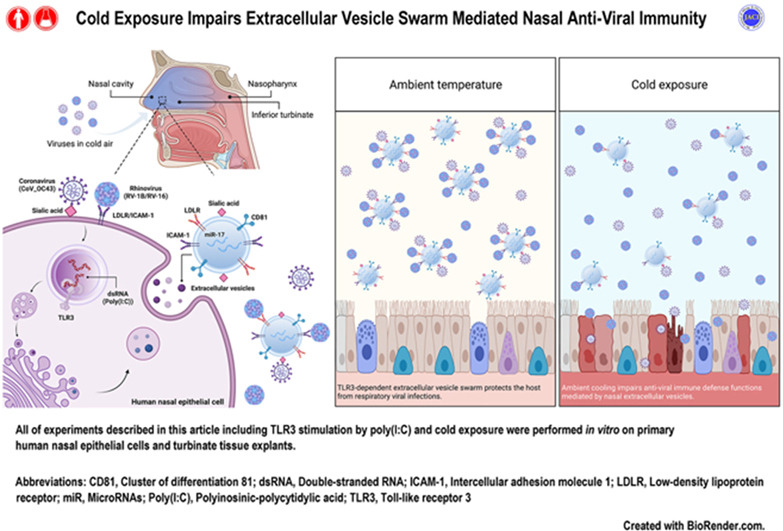
Background The human upper respiratory tract is the first site of contact for inhaled respiratory viruses and elicits a series of innate immune responses. The seasonal variation of respiratory viral infections and the importance of ambient temperature in modulating immune responses to infections have been well recognized; however, the underlying biological mechanisms remain unstudied. Aim We investigated the role of extracellular vesicles (EVs) derived from nasal epithelium in Toll-like receptor 3 (TLR3)-dependent innate antiviral immunity. Methods We evaluated the secretion and composition of nasal epithelial EVs after TLR3 stimulation in human autologous cells and fresh human nasal mucosa surgical specimens. We also explored the antiviral activity and mechanisms of TLR3-stimulated EVs against respiratory viruses, as well as the effect of cold ambient temperature on TLR3-dependent antiviral immunity. Results We found that exposure to polyinosinic:polycytidylic acid, also known as poly(I:C), induced a swarm-like increase in nasal epithelial EV secretion through TLR3 signaling. EVs participated in TLR3-dependent antiviral immunity, protecting the host from viral infections through EV-mediated functional delivery of miR-17 and direct neutralization of the virion after binding to virus ligands via receptors. including LDLR and ICAM-1. These potent antiviral immune defense functions mediated by TLR3-stimulated EVs were affected by cold exposure through a decrease in total EV secretion as well as a decrease in microRNA packaging and antiviral binding affinity of individual EVs. . Conclusion TLR3-dependent nasal epithelial EVs exhibit multiple innate antiviral mechanisms to suppress respiratory viral infections. Furthermore, our study provides a direct quantitative mechanistic explanation for the seasonal variation in the prevalence of upper respiratory tract infections. |
Comments
Colder temperatures suppress a newfound immune response inside the nose, offering evidence why colds, flu and COVID-19 are more common in colder months
Researchers at Mass Eye and Ear and Northeastern University have discovered a previously unidentified immune response inside the nose that fights viruses responsible for upper respiratory infections. Additional testing revealed that this protective response is inhibited in colder temperatures, making infection more likely to occur.
The new study, published in The Journal of Allergy and Clinical Immunology , offers the first biological mechanism to explain why viruses like the common cold, flu and COVID-19 are more likely to increase in colder seasons, according to researchers. authors.
“Cold and flu season was conventionally thought to occur in colder months because people were more stuck indoors where airborne viruses could spread more easily,” said Benjamin S. Bleier, MD, FACS, director. of Otolaryngology Translational Research in Mass Eye and Ear. and lead author of the study. “However, our study points to a biological cause for the seasonal variation in upper respiratory viral infections we see each year, most recently demonstrated during the COVID-19 pandemic.”
First line defense on the nose
The nose is one of the first points of contact between the outside environment and the inside of the body and, as such, a likely entry point for disease-causing pathogens.
Pathogens are inhaled or deposited directly (such as by hands) in the front of the nose, where they make their way back through the airways and into the body infecting cells, which can lead to a yeast infection. the upper respiratory tract. How the respiratory tract protects itself against these pathogens has long been poorly understood.
That was until a 2018 study led by Dr. Bleier and Mansoor Amiji, PhD, Distinguished Professor of Pharmaceutical Sciences at Northeastern University, discovered an innate immune response that is triggered when bacteria are inhaled through the nose: the cells in the The front part of the nose detected the bacteria and then released billions of small fluid-filled sacs called extracellular vesicles (or EVs, formerly known as exosomes) into the mucus to surround and attack the bacteria. Dr. Bleier compares unleashing this swarm of EVs to “kicking a hornet’s nest.”
The 2018 study also showed that EVs carry protective antibacterial proteins through mucus from the front of the nose to the back along the airways, which then protects other cells against the bacteria before penetrate too much into the body.
For the new study, the researchers sought to determine whether this immune response was also triggered by viruses inhaled through the nose, which are the source of some of the most common upper respiratory infections.
Virus fighting mechanism tested in various conditions
Led by the study’s first author, Di Huang, PhD, a researcher at Mass Eye and Ear and Northeastern, the researchers analyzed how cells and nasal tissue samples collected from the noses of patients undergoing surgery and healthy volunteers responded to three viruses. : a single coronavirus and two rhinoviruses that cause the common cold.
They found that each virus triggered an EV swarm response from nasal cells, although using a different signaling pathway than that used to fight bacteria. The researchers also discovered a mechanism at play in the response against viruses: upon release, the EVs acted as decoys, carrying receptors to which the virus would bind instead of nasal cells.
“The more decoys, the more EVs can absorb viruses into the mucus before the viruses have a chance to bind to nasal cells, suppressing infection,” Dr. Huang said.
The researchers then tested how colder temperatures affected this response, which is especially relevant in nasal immunity given that the internal temperature of the nose is highly dependent on the temperature of the outside air you inhale. They took healthy volunteers from a room-temperature environment and exposed them to temperatures of 4.4°C (39.9°F) for 15 minutes and found that the temperature inside the nose dropped by about 5°C. They then applied this temperature reduction to the nasal tissue. samples and observed an attenuated immune response. The amount of EVs secreted by the nasal cells decreased by almost 42 percent and the antiviral proteins in the EVs were also affected.
“Combined, these findings provide a mechanistic explanation for the seasonal variation in upper respiratory tract infections,” Dr. Huang said.
Therapeutic potential
Future studies will aim to replicate the findings with other pathogens. The studies could be carried out as challenge studies, where an animal or human model is exposed to a virus and its nasal immune response is measured.
From their recent findings, the researchers can also imagine ways the therapy may induce and strengthen the nose’s innate immune response. For example, a drug therapy, such as a nasal spray, could be designed to increase the number of EVs in the nose or binding receptors within vesicles.
“We have discovered a new immune mechanism in the nose that is constantly being bombarded and we have shown what compromises this protection,” said Dr. Amiji. "The question now changes to, ’How can we take advantage of this natural phenomenon and recreate a defensive mechanism in the nose and increase this protection, especially in the colder months?’"