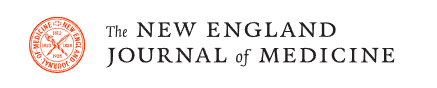Highlights
|
The METEOR-China randomized controlled trial (RCT) demonstrated that rosuvastatin 20 mg/day significantly reduced rates of carotid intima-media thickness (cIMT) progression compared with placebo over a 2-year period in Chinese adults with subclinical atherosclerosis.
Statins exert a beneficial effect on cIMT progression and rates of stroke/cardiovascular events in high-risk patients (eg, those with coronary heart disease 6 or recent stroke/transient ischemic attack).
Background
Atherosclerosis is the leading cause of cardiovascular disease worldwide, including in China. Primary prevention, through lipid reduction, could prevent the development of atherosclerosis. Carotid intima-media thickness (CIMT) is a well-validated measure of atherosclerosis used in intervention studies as a primary outcome and surrogate endpoint for cardiovascular disease events.
Methods
This randomized, double-blind, placebo-controlled, multicenter, parallel-group study evaluated the effects of rosuvastatin 20 mg/d compared with placebo on CIMT progression over 104 weeks in Chinese people with subclinical atherosclerosis.
The primary endpoint was the annualized rate of change in the mean of maximum CIMT measurements taken 7 times during the study period from each of the 12 carotid artery sites (near and far walls of the right and left, carotid bulb and internal carotid artery). carotid artery).
Secondary endpoints included changes in CIMT at different arterial sites and changes in lipid parameters. Safety was also evaluated.
Results
Participants were randomized (1:1) to receive rosuvastatin (n=272) or placebo (n=271). Baseline characteristics were well balanced between groups.
The change in mean maximum CIMT of the 12 carotid sites was 0.0038 mm/a (95% CI, -0.0023-0.0100) for the rosuvastatin group versus 0.0142 mm/a ( 95% CI, 0.0080-0.0204) for the placebo group, with a difference of -0.0103 mm/year (95% CI, -0.0191 to -0.0016; P = 0.020) .
For CIMT secondary endpoints, results were generally consistent with the primary endpoint. There were clinically relevant improvements in lipid parameters with rosuvastatin. We observed an adverse event profile consistent with the known safety profile of rosuvastatin.
Conclusions
Rosuvastatin 20 mg/d significantly reduced CIMT progression over 2 years in Chinese adults with subclinical atherosclerosis and was well tolerated.