Effect of Continuous Infusion of Hypertonic Saline Versus Standard Care on Neurological Outcomes at 6 Months in Patients With Traumatic Brain Injury The COBI Randomized Clinical Trial
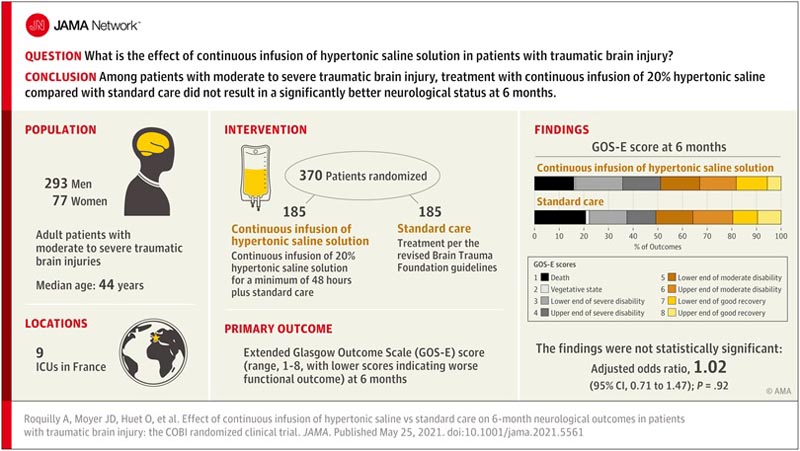
Key points Question What is the effect of continuous infusion of hypertonic saline in patients with head trauma? Findings In this randomized clinical trial that included 370 adults with moderate to severe traumatic brain injury, treatment with continuous infusion of 20% hypertonic saline versus standard care resulted in an odds ratio for better neurological outcomes (according to Extended Glasgow Outcome Scale) of 1.02 after 6 months; this was not statistically significant. Significance Among patients with moderate to severe traumatic brain injury, treatment with continuous infusion of 20% hypertonic saline compared with standard care did not result in significantly better neurological status at 6 months. |
Importance
Fluid therapy is an important component of care for patients with traumatic brain injury, but it is unclear whether it modulates clinical outcomes.
Aim
To determine whether continuous infusion of hypertonic saline improves neurological outcome at 6 months in patients with traumatic brain injury.
Design, environment and participants
Multicenter randomized clinical trial conducted in 9 intensive care units in France, including 370 patients with moderate to severe traumatic brain injury who were recruited between October 2017 and August 2019. Follow-up was completed in February 2020.
Interventions
Adult patients with moderate to severe traumatic brain injury were randomly assigned to receive a continuous infusion of 20% hypertonic saline plus standard care (n = 185) or standard care alone (controls; n = 185).
20% hypertonic saline was administered for 48 hours or longer if patients remained at risk for intracranial hypertension.
Main results and measures
The primary outcome was the Glasgow Outcome Scale-Extended (GOS-E) score (range, 1-8, with lower scores indicating worse functional outcome) at 6 months, obtained centrally by blinded raters and analyzed with ordinal logistic regression adjusted for prespecified prognostic factors (with a common odds ratio [OR] > 1.0 favoring intervention).
12 secondary outcomes were measured at multiple time points, including the development of intracranial hypertension and 6-month mortality.
Results
Among the 370 patients who were randomized (median age, 44 [interquartile range, 27-59] years; 77 [20.2%] women), 359 (97%) completed the trial. The adjusted common OR for GOS-E score at 6 months was 1.02 (95% CI, 0.71-1.47; p = 0.92).
Of the 12 secondary outcomes, 10 were not significantly different. Intracranial hypertension developed in 62 (33.7%) patients in the intervention group and 66 (36.3%) patients in the control group (absolute difference, −2.6% [95% CI, −12 .3% to 7.2%]; OR, 0.80 [95% CI, 0.51-1.26]).
There was no significant difference in 6-month mortality (29 [15.9%] in the intervention group versus 37 [20.8%] in the control group; absolute difference, -4.9% [CI 95%, -12.8% to 3.1%]; hazard ratio, 0.79 [95% CI, 0.48-1.28]).
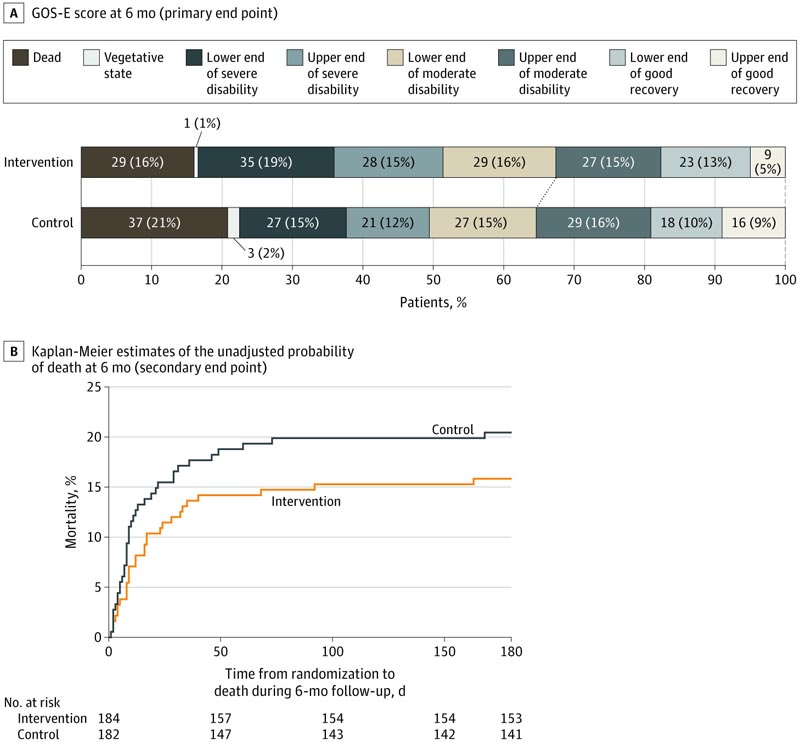
A, Distribution of Glasgow Outcome Scale-Expanded (GOS-E) scores at 6 months. The different colors correspond to the GOS-E scores. The connecting line between the 2 study groups indicates dichotomization of GOS-E results (poor vs. favorable). B, Kaplan-Meier estimates of the unadjusted probability of death at 6 months in patients receiving continuous infusion of 20% hypertonic saline or standard care. The estimated adjusted probability of death at 6 months is a hazard ratio of 0.79 (95% CI, 0.48-1.28). The median observation time was 180 days (interquartile range, 180-180 days) in both treatment groups. The graphic evaluation indicates that the proportionality assumption was met.
Conclusions and relevance Among patients with moderate to severe traumatic brain injury, treatment with continuous infusion of 20% hypertonic saline compared with standard care did not result in significantly better neurological status at 6 months. However, the confidence intervals for the findings were wide and the study may have had limited power to detect a clinically important difference. |
ClinicalTrials.gov Trial Registry Identifier: NCT03143751